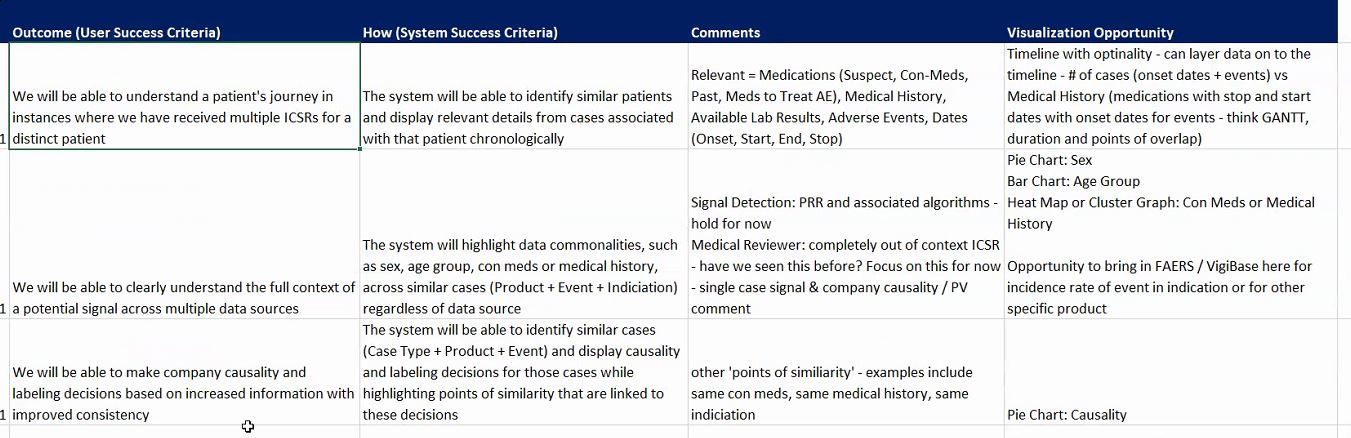
Last month, Cambridge Semantics held its inaugural Life Sciences Smart Data Discovery and Analytics Forum in Boston. At this forum, leaders in Life Sciences and Big Data met to discuss the...
With CDISC Adoption by FDA, Managing Clinical Trial Metadata with Semantic Technology Has Emerged Front and Center!
As FDA outlines, “the submission of standardized study data enhances a reviewer’s ability to more fully understand and characterize the efficacy and safety of a medical product,” and adopted CDISC...